 Early atomic mass theory was proposed by the English chemist William Prout in a series of published papers in 18. The first scientists to measure atomic mass were John Dalton (between 18) and Jons Jacoband Berzelius (between 18). So 1 u is 1/12 of the mass of a carbon-12 isotope: Both units are derived from the carbon-12 isotope, as 12 u is the exact atomic mass of that isotope. The atomic mass is usually measured in the units unified atomic mass unit (u), or dalton (Da). These concepts are further explained below. Mass of molecule calculated from the mass of its isotopes (in contrast of measured ba a mass spectrometer) Integer mass of molecule consisting of most abundant isotopes Ratio of mass m of a molecule and and the atomic mass constant m u Ratio of mass m and and the atomic mass constant m u Mass Concepts in Chemistry name in chemistry Note that the former is now often referred to as the "molecular weight" or "atomic weight". The former usually implies a certain isotopic distribution, whereas the latter usually refers to the most common isotope ( 16O 2). For example, the macroscopic mass of oxygen (O 2) does not correspond to the microscopic mass of O 2. This means that from a physical stand point, these mixtures are not pure. On the macroscopic level, most mass measurements of pure substances refer to a mixture of isotopes. In addition, the situation is rendered more complicated by the isotopic distribution. The name "atomic mass" is used for historical reasons, and originates from the fact that chemistry was the first science to investigate the same physical objects on macroscopic and microscopic levels. "Neither the name of the physical quantity, nor the symbol used to denote it, should imply a particular choice of unit." Later, after heating, the molecule releases a molecule of carbon dioxide (CO 2).\) The organic molecule cyclobutadiene (C 4H 6) combines with propenoic acid (C 3H 4O 2) to form a molecule with the molecular formula of C 7H 10O 2. You have of some unknown transition metal which weighs. 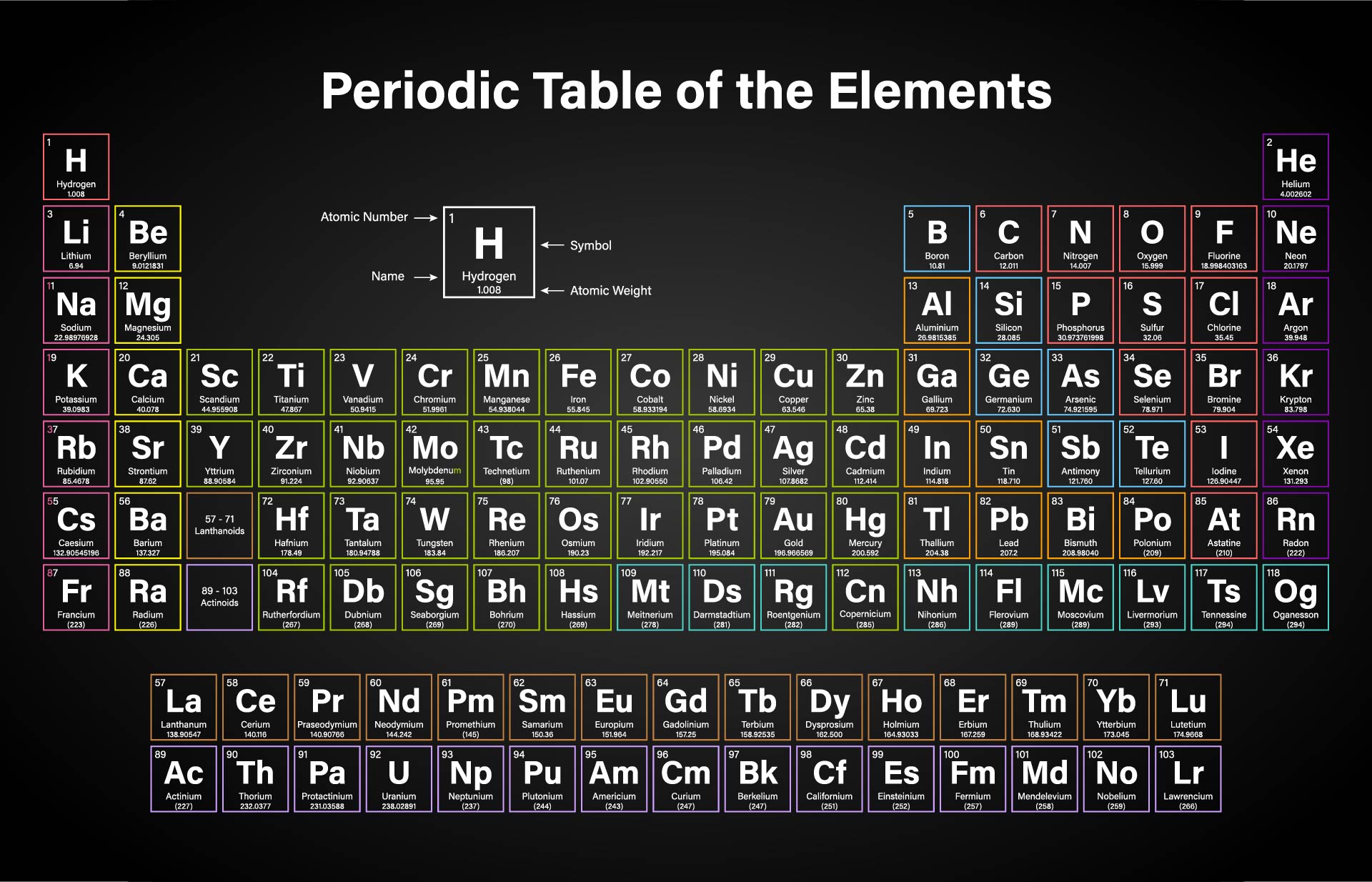
Nitrogen and hydrogen: Carbon Dioxide (CO 2) Example Problem 
Moreover, this means that of NH 3 is equal to of NH 3. For ammonia, we should therefore get a molecular mass of. The last and final step is to add up the two products we got from multiplying. So, now we will multiply these numbers by their corresponding atomic masses. 
According to the molecular formula (NH 3), there is one nitrogen and three hydrogens present. The second step is to determine how much of each element is present in the compound. For example, if we are trying calculate for ammonia (NH 3), then we need to find the atomic masses for nitrogen and hydrogen. The atomic mass is equal to the atomic number which is listed below the element symbol. The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. Let’s look at some examples of calculating this value for some different molecules. It should be noted that this number is an average and therefore may vary due to isotopic elements. Molar mass can be calculated by using the periodic table and following three simple steps. The units of molar mass is grams per mole, often abbreviated. That’s a heck of a lot of molecules, which is why the molecular mass of table salt (sodium chloride) is a respectable 58.44 grams per mole, quite a large handful. Both definitions will give you the same result, they are just in different units.Ī “mole” of a substance is defined as atoms or molecules of that substance. Another definition is the sum of the atomic weights of the atoms that make up a molecule. The definition of molar mass is simply the number of grams that one mole of a substance weighs. Molecular formula: A way of presenting a molecule that shows the proportions of atoms.Molecules: Neutral group of atoms chemically bonded together.Compounds: Contains atoms of different elements that are combined at a fixed ratio.It is sometimes referred to as molecular weight. Molar mass: The sum of atomic weight in a molecule.Isotope Abundance and Average Atomic Mass.How to Write Electron Shell Configurations. /PeriodicTableSigFig-NoBG-56a12da75f9b58b7d0bcd00f.png)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
